ADVANCING SUSTAINABILITY IN AQUACULTURE

Mitigating ocean acidification with marine algae
Kelp is an emerging aquaculture star with uses for direct consumption, health additives and feed for livestock. We are looking into the potential of sugar kelp to mitigate ocean acidification effects on larval fish as well as benefit larval gut microbiota. This has applications for habitat restoration for wild populations, as well as uses in multi-trophic aquaculture to improve sustainability and fish welfare.
Sustainable feeds
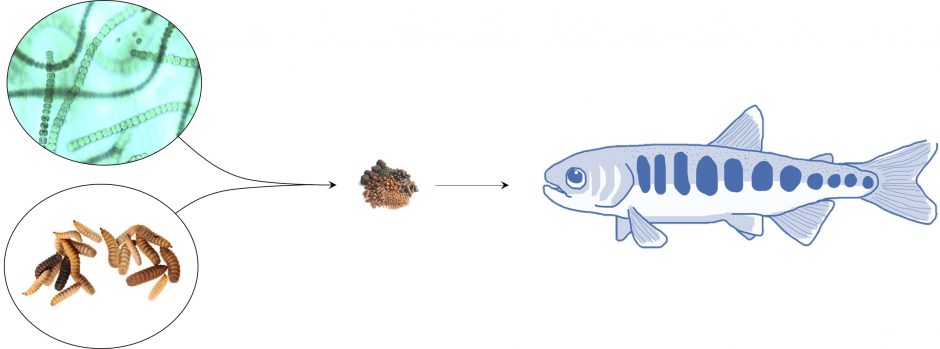
Feed is the largest input cost for most aquaculture operations. Aquaculture growth and sustainability issues are driving demand for alternatives to fishmeal. Insect-based feeds offer a sustainable, healthy alternative but the application to the early life stages unclear. For example, larvae have ~2x higher omega-3 fatty acid requirement for development.
In collaboration with industry and DFO, we are currently launching a project to research sustainable feed alternatives on digestive development and growth in juvenile fish, as well as uses of humic and fulvic acids to improve the immune system.
ENVIRONMENTAL STRESSORS AND THEIR EFFECTS ON COMMERCIAL FISH

In controlled experiments, we manipulate CO2, temperature, oxygen and food to understand the effects of combined climate change stressors on the physiology early life stages of commercial fish.
CO2 effects on commercial fish larvae
Elevated CO2 concentrations, such as occurring with ocean acidification, but also common in high density rearing environments and recirculating aquaculture systems can be problematic for early stages of fish. We are researching species- and life stage specific effects of CO2 on growth, metabolism and development in commercial fish, such as Atlantic cod, Atlantic herring and yellowfin tuna.
These results are used in stock assessment models and population dynamics models such as SEPODYM that integrate climate change with fisheries data. In aquaculture, this data is used for production and welfare management in hatcheries.
Recent publications:

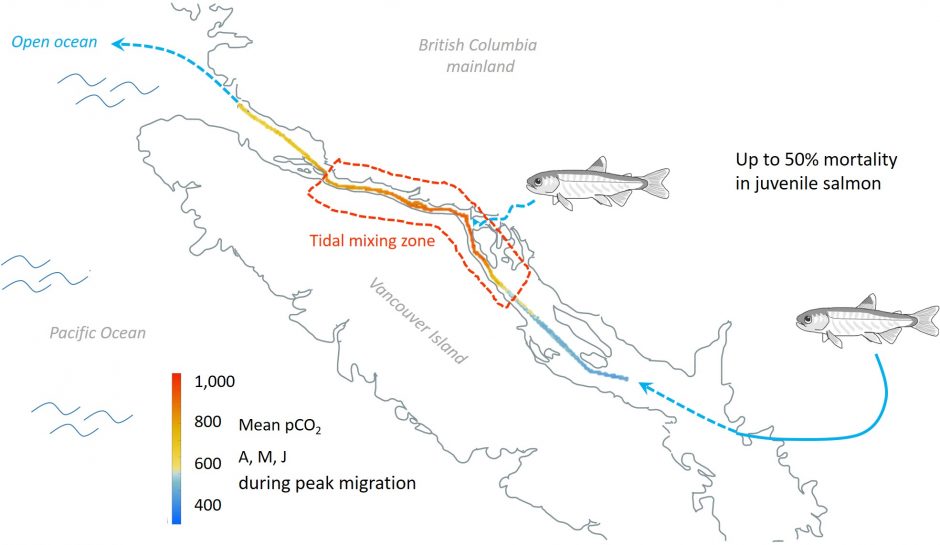
Combined climate change stressors and juvenile salmon
Populations of many wild juvenile Pacific salmon off the coast of Southern BC have experienced drastic declines. Direct and indirect effects of climate change are likely a contributing factor to the high mortality in outmigrating juveniles. Additionally, many salmon net pens are located in this area, subjecting fish to the same oceanic conditions.
In collaboration with the Hakai Institute, we are researching how challenging ocean conditions affect their growth, acid-base regulatory capacity, condition and immune defense in juvenile salmon.
Current and forthcoming publications:
Frommel, A.Y., Brauner, C.J., Hunt, B.P.V. Effects of ocean acidification and warming on smoltification success in chinook salmon (in prep.)
Frommel, A.Y., Lye, S., Collicutt, B., Hunt, B.P.V. Combined effects of ocean acidification and food limitation on condition of juvenile chum salmon (in prep.)
LOCAL VARIABILITY AND RESILIENCE TO CLIMATE CHANGE
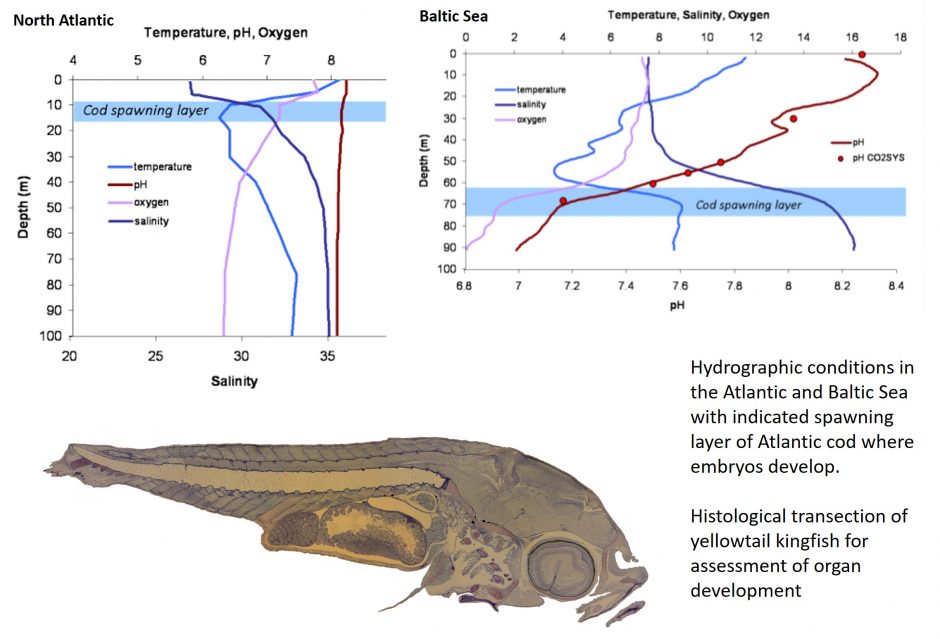
Are populations living in naturally CO2 enriched regions resilient to ocean acidification?
We compare populations living in naturally CO2 enriched areas to “naive” populations to determine the adaptation potential of populations to climate change. One example of this is cod living in the North Atlantic were found to be vulnerable to ocean acidification, whereas cod from the Baltic population that develop in naturally high CO2 water layers were resilient. Whether this is a common trend remains to be determined, however studies on other species suggest this may not be the case. Yellowfin tuna larvae, that develop in upwelling systems off the coast of Panama were found to be vulnerable to high CO2 levels, whereas yellowtail kingfish, which develop in low CO2 coastal regions off the coast of New Zealand, were not. This suggest yellowtail kingfish may be a suitable species for further investment for fisheries and aquaculture.
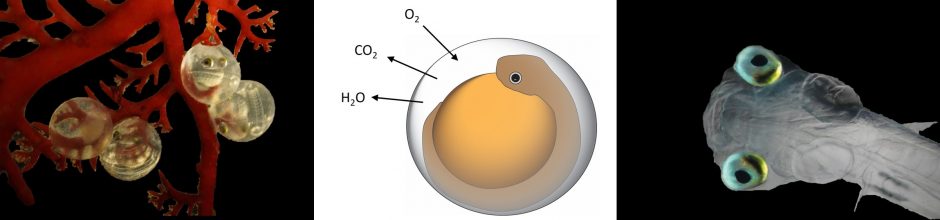
Air exposure and CO2 tolerance
CO2 is has been demonstrated to negatively affect survival, growth and organ development in herring larvae. We postulate that air exposure during low tide in intertidally spawned herring eggs may allow embryos to offgas excess CO2 and develop normally.
Media coverage: https://hakai.org/a-fish-egg-out-of-water/
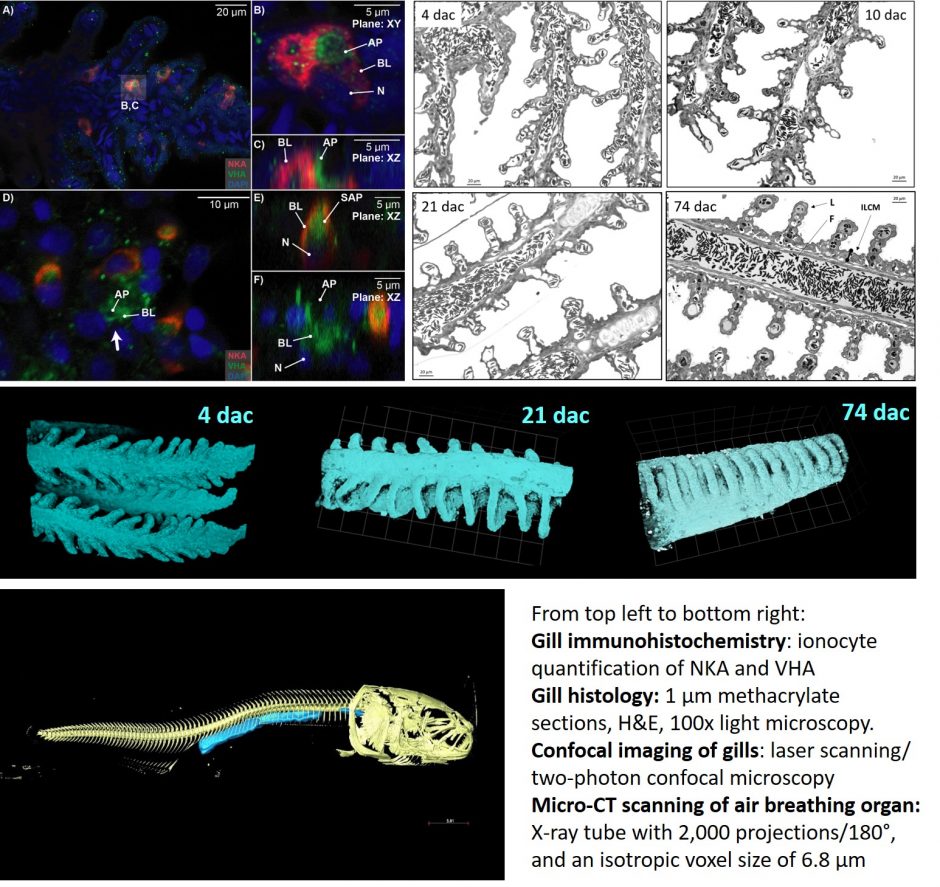
Living in extremes: Arapaima gigas
Arapaima gigas, one of the world’s largest freshwater fish and an important aquaculture species in Brazil, lives in oxygen- and ion-poor waters of the Amazon River System. To thrive during seasonal hypoxia, A. gigas develop into obligate air-breathers as juveniles. This project investigates the transition from water to air-breathing juvenile Arapaima gigas undergo and the associated modifications to the gills and the swim bladder into an air-breathing organ.
Respiration and ion-regulation capacity are analyzed with a variety of methods.
The gas diffusion distance in the gills and air-breathing organ are quantified using histology. The surface area of the gills are determined with confocal imaging and the surface area of the air-breathing organ via micro-CT scanning. Ion regulatory capacity at the gills is quantified using immunihistochemistry.